|
To truly understand why space science plays such an important role in our everyday lives, we must answer a simple, but fundamental question: “What is the Sun?“ From Encyclopædia Britannica, “sun” is defined as “star around which Earth and the other components of the solar system revolve“. Okay, sounds right, but then what exactly is a star? Again from Encyclopædia Britannica, “star“ is “any massive self-luminous celestial body of gas that shines by radiation derived from its internal energy sources“. So, putting those two definitions together we could say that the Sun is “the massive self-luminous celestial body of gas that shines by radiation derived from its internal energy sources around which Earth and the other components of the solar system revolve“. Simple, right? Let’s break that down a bit, starting with the simplest part, the end. The Sun is the center of our solar system and all of the planets, including the Earth, revolve or orbit around it. Of course, we didn’t always know or believe that, but that’s a story for a different post. Heading back to the beginning of the definition, we see the word “massive“. Massive here is used in the most literal meaning of the word. The Sun is by far the most significant object in our solar system, making up more than 98% of the total mass. It’s 333,000 times more massive than the Earth, meaning it is made up of that much more matter. In literal dimensions, you could line up more than 100 Earths across the equator of the Sun and fit over 1 million Earths inside the Sun. That’s pretty huge when we think on a human or even a global scale, but keep in mind that the Sun is just a mid-size star! So, next we’ll focus on the “body of gas” part of the definition. So, in elementary school you probably learned that there are three forms of matter: solid, liquid, and gas. Well, that’s not entirely true (“gasp“), there’s actually a fourth state of matter known as plasma. Really, it can be considered a special type of gas, an ionized gas, but since that ionization causes it to act in ways that are very different than a regular gas, it should be (and usually is) considered its own state of matter. Unlike regular gases, plasmas are electrically conductive and respond to electromagnetic forces. You’re probably extremely familiar with plasmas and their applications, without even realizing it: lightning, electric sparks, fluorescent and neon lights, and some TVs. So the Sun is actually a body of ionized gas (or plasma). Finally, the most important parts of the definition that truly define a star are “self-luminous” and “shines by radiation derived from its internal energy sources“. These statements refer to the fact that the Sun and other stars actually produce their own light (or radiation), unlike planets and the Moon which simply reflect light from the Sun. So what are these internal energy sources that are mentioned? Well, the inner cores of stars are the only natural environments (that we know of) where it’s hot enough (27,0000,000° F) and high enough density (150 g/cm³, roughly 10 times denser than lead) for nuclear fusion to take place. Nuclear fusion is the combination of smaller atoms of elements to make larger ones. In the cores of stars like our Sun, the lightest element, hydrogen (H), is converted into the second lightest element, helium (He), in a three-step process known as the proton-proton or “pp” chain” (cue giggling). 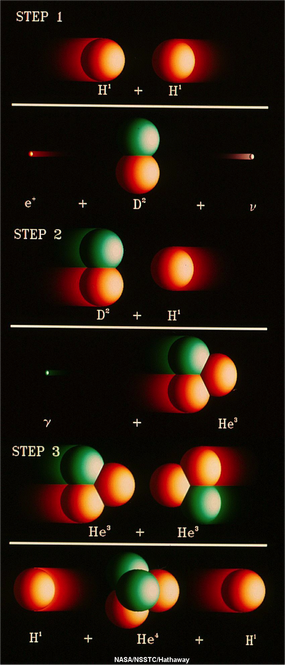 This image shows the steps of the proton-proton (pp) chain, the nuclear fusion reaction that converts hydrogen (H) into helium (He) in the core of the Sun. In the first step two protons collide to produce deuterium, a positron, and a neutrino. In the second step a proton collides with the deuterium to produce a helium-3 nucleus and a gamma ray. In the third step two helium-3s collide to produce a normal helium-4 nucleus with the release of two protons. Image credit: NASA In this reaction, four hydrogen atoms are converted into a single helium atom. That sounds all well and good, but it doesn’t quite explain why that conversion results in the massive energy production that we all know occurs in the Sun. The secret to that comes from what is probably the most famous equation in all of science: the mass-energy equivalence equation (E=mc²). You’ve probably seen that equation once or twice, and you probably know that it was introduced by arguably the most famous scientist ever Albert Einstein, and you probably incorrectly think that that’s why Einstein won the 1921 Nobel Prize in Physics (he actually won it for a different discovery he made in 1905, something known as the photoelectric effect). Anyway, this simple equation is the reason why the Sun is the most powerful energy source known to man. Remember those four little hydrogen atoms? The combined mass of those four hydrogen atoms (4 x 1.6605402 x10–27 kg = 6.64187×10–27 kg) is actually less than the mass of the one new helium atom ( 6.64648×10–27 kg). Note that the difference is miniscule, a mere 4.61×10-30 kg, but looking at Einstein’s equation, that tiny mass is multiplied by a really large number, the speed of light squared (8.98755179×1016 m2/s2). That means that the total energy released by each pp chain is 4.14326137×10-13 kg·m2/s2 (or Joules). That’s 1×10-19kW·h, the basic unit by which electric companies charge for usage. So each reaction is actually liberating only a tiny bit of energy, but when you multiply that by the enormous amount of mass in the Sun and mind-boggling number of reactions that are occurring in the core, you can see the energy add up. But the really crazy fact is that even once the light is emitted from the core in the form of a photon, it takes tens or even hundreds of thousands of years (estimates range from 17,000 to 1 million) for the photon to actually reach the surface of the Sun and escape out into space. That’s because the photon can be randomly emitted in any given direction, not necessarily towards the surface. Then, because the Sun is so dense, it doesn’t travel very far before the photon bumps into another atom and is absorbed. Then that photon is emitted again in a random direction. This all adds up to a really, really long “random walk” through the Sun to the surface.
This post was originally written by Ian Cohen for his blog "At the Center of It All"
0 Comments
 This year seems to be a year of action. Standing up for scientists being able to freely share their ideas and results, standing up for scientists being allowed to go to conferences and to collaborate, and of course standing up for civil liberties and the values that make us a wonderful country. Along with this, there have been quite a few opinion pieces in some of the big news organizations about whether or not science and scientists should be political (Vox.com The New York Times Forbes Slate The Washington Post and many more). Science has always been political, if you've ever been to a faculty meeting, organized a conference, or even just talked with colleagues over beers, you can see/hear about some of the politics in science. Granted this isn't what the news articles were referring to. So the questions stands, should science and really scientists be involved and speak about politics?
I think there are two answers, so lets take the easiest first. Should science be political? No. Science, the act of researching and presenting results should not be political... well except when the experiments involve living beings or could hurt living beings, but that's more about ethics and regulations on types of experiments allowed. But the research, the data collected, the communication of results through publications and conferences should not be silenced or manipulated for a persons or funding agencies own political views. Now I'm sure it's not quiet so black and white and a bit more grey then I've written here, but lets move on to the scientists. Should scientists be involved in politics? Yes! At the very least, only with informed decisions can we have good policy. The ultimate end goal of science is to understand the world around us and help improve our lives in it. Often this means that science and research is seen as turning into technology or medical advances, but it also leads to policy and regulations. Take for instance seismology. Understanding how earthquakes propagate, and how large they are likely to be has informed building codes. Or how about my beloved space weather? Well, there's a space weather research and forecasting bill introduced to the senate right now looking at understanding what the effects are on power grids and oil pipelines as well as on aviation and many other aspects of our lives. And of course there is the big one, climate science.... but I don't think we need to go into that here. Since much of science can inform policy, we need scientist to be involved in politics and to communicate this information. Scientists also need to be involved in politics to make sure that this pipeline stays open. We saw earlier this year how the government felt that it was appropriate and perfectly fine to gag scientists, not allowing them to speak as well as "reviewing" the data and results before they went public. Sure we sometimes have to do this to make sure the research is not classified or competition sensitive, but what they were starting went well beyond this, removing already public data. The government also make policies which effect scientists! For instance here's a list of some of the current and recently passed legislation that could effect my field: 1) The Space Weather Research and Forecasting act 2) The Inspiring the Next Space Pioneers, Innovators, and Researchers and Explorers (INSPIRE) Women Act 3) To direct NASA to return humans on the moon and to develop a sustained human presence on the moon 4) A manifest for human space flight And many many more. Some dictating the way lab waste is treated, others discussing what federal regulations contractors have to follow or not follow, and a few more on humans in space or going to the moon or mars. Many if not all have been advised at some point by scientists. And scientists should be commenting on them all! Anyone with an interest in space weather research should read and write/call your representatives about them! And for the last and most important reason... Which really should be self explanatory... Scientists are citizens and it is our civic duty to be involved in our government. This post has been re-published with permission from AlexaHalford.weebly.com  Name: Ian Cohen Institution (Location): The Johns Hopkins University Applied Physics Laboratory (Laurel, MD) Degree(s): Ph.D., Physics, University of New Hampshire, 2015; B.A., Astronomy & Physics, Boston University, 2010 Current and past missions/projects: NASA Magnetospheric Multiscale (MMS), NASA sounding rocket program (RENU2, MICA, ACES missions) Research Interests: Plasma physics; magnetosphere-ionosphere coupling (specifically ion outflow, auroral dynamics, and ionospheric feedback); energetic particle dynamics; plasma and energetic particle instrumentation
|
AuthorA bunch of us Space Nerds Archives
July 2017
Categories
All
|

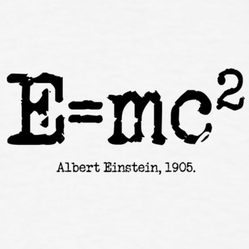
 RSS Feed
RSS Feed
